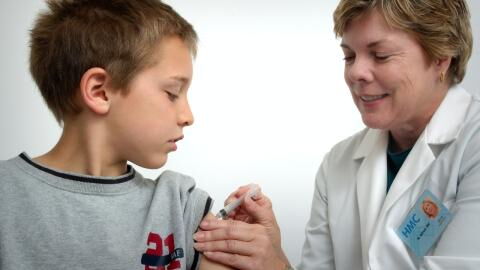
It is normal and expected to feel discomfort after injecting a COVID-19 vaccine. As soon as the serum enters your arm, your blood flow increases and your immune system rushes on the virus. This can result in pain around the injection site, the most common side effect of the three vaccines licensed in the United States. The reaction is more common after the Pfizer and Moderna vaccines than that of Johnson & Johnson.
Discover our latest podcast
Nothing too serious
Less than 50% of participants in the Johnson & Johnson clinical trial reported injection site pain, compared to 92% of Moderna participants and 84% of Pfizer participants. In the case of AstraZeneca, a vaccine authorized in Europe but not in the United States, sensitivity at the injection site was the most common, affecting 64% of trial participants. About 54% of participants reported pain at the injection site.
When our immune system detects the ingredients of a vaccine, it also releases inflammatory chemicals to protect us. This is why some patients have fever, muscle pain, severe fatigue or headache soon after the injection.
Fatigue was the second most common side effect in the Moderna and Pfizer trials. Almost 69% of Moderna adherents and 63% of Pfizer adherents reported it. But headaches were slightly more common than fatigue among Johnson & Johnson participants: 39% reported headaches, compared with 38% who reported fatigue. In the AstraZeneca trial, fatigue and headaches were just as common: around 53% of participants said they had such effects.
Data from Pfizer, Moderna and Johnson & Johnson is based on clinical trials and AstraZeneca data is based on a study of 130 people.
Fatigue and headaches are more common after the second dose of Pfizer and Moderna, but not after an injection of AstraZeneca
A recent report looked at side effects in more than 1.9 million people who received both doses of Pfizer or Moderna vaccines. Overall, side effects were slightly more common after the two doses of Moderna than after the two doses of Pfizer. And the side effects were more numerous and more serious after the second dose of either vaccine.
Patients with local pain after injection went from 68% after the first dose of either vaccine to 72% after the second dose. Fatigue increased from 31% to 54% between the first and second doses, and headaches from 26% to 47%. Almost 82% of patients who received the Moderna serum reported some local reaction (pain, redness, itching or swelling) after their second dose. 69% of patients receiving a dose of Pfizer had the same skin reaction. Additionally, 60% of Moderna recipients reported fatigue and 53% reported headache after the second dose. While for the second dose of Pfizer, 48% of people vaccinated reported fatigue and 40% headaches.
The AstraZeneca vaccine tends to have stronger side effects after the first dose, for reasons scientists do not fully understand. Experts say the reaction could be linked to the vaccine's technology, which uses a genetically engineered cold virus to introduce a coronavirus gene into the body. This cold virus could potentially stimulate a stronger immune response from the start.
Muscle aches and pains are more common than stomach problems
Muscle pain was among the most common side effects in all four trials. In Moderna's, 60% of participants experienced muscle aches, while 38% of Pfizer participants reported this symptom. About a third of Johnson & Johnson participants and 44% of AstraZeneca participants also reported this kind of pain.
Chills were less common but not uncommon: 43% of people in the Moderna trial complained of it, as did 32% of participants in the Pfizer study and the AstraZeneca study. Only 2% of Johnson & Johnson participants felt this effect. In the Pfizer and Moderna trials, 15% of participants reported fever, compared to 9% in the Johnson & Johnson trial and 8% in the AstraZeneca trial.
Overall, gastric problems such as nausea, vomiting, and diarrhoea were not commonly associated with Pfizer or Moderna vaccines, but about 14% of Johnson & Johnson participants and 22% of AstraZeneca participants have reported some.
An overall short experience
Adverse effects were short-lived in all four trials. The majority of Moderna participants said they started on the day of the injection and lasted two days after each odose. On average, Pfizer participants also experienced side effects one to two days after their injection, with the reaction usually lasting only one day.
Johnson & Johnson participants experienced side effects within two days of their injection. On average, the fatigue, headaches, and muscle aches lasted for two days, while nausea and fever lasted for a day. For patients who received the AstraZeneca serum, side effects usually subsided within a few days as well.